Research
Experimental models of antibody-driven CNS autoimmunity with a focus on visual outcomes
With advancements in autoantibody detection methods and the identification of antibodies of pathogenic relevance, a better differentiation of MS from autoantibody-mediated demyelinating CNS syndromes such as NMOSD and MOG-AD has become possible. Still, the distinct features of their immunopathology are insufficiently understood.
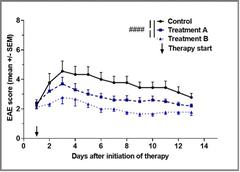
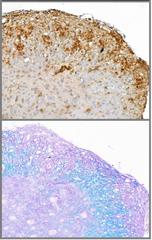
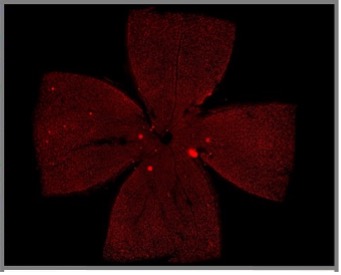
With visual impairment representing a major symptom of MS, NMOSD and MOG-AD severely affecting the patient’s daily living and quality of life, the better understanding of its pathophysiology is crucial. Moreover, the anterior visual system serves as a very suitable model to assess pathophysiological features of demyelinating CNS disorders as it is an easily accessible neuronal structure, known to be involved in different entities. This structure can be measured on a functional and structural level in a standardized way, not only in the human disease, but also in animal models. In particular, neuronal degeneration is possibly reflected in retinal degeneration with axonal and ganglion cell loss and measureable via optical coherence tomography (OCT). In a murine experimental model system mimicking MOG-AD, we successfully demonstrated a positive treatment effect of a novel monoclonal antibody directed against the neonatal Fc receptor (FcRn) on both spinal cord and visual disease manifestations (funded by UCB Pharma). This treatment approach is currently being transferred into a phase 3 treatment study for patients with MOG-AD.
Understanding of steroid signaling in people with neuroimmunological diseases
Glucocorticosteroids (GC) are the mainstay of relapse treatment, however, approximately one third of our patients do not fully recover, leading to an increased disability. We have recently demonstrated that vitamin D deficiency contributes to GC resistance, as it is associated with a downregulation of the GC receptor. Findings were consistent in a mouse model of MS and in human T cells, thus providing a possible target for increasing the efficacy of GC therapy in neuroimmunological diseases. An interplay between vitamin D and GC and their respective receptors has been demonstrated which is currently further investigated, taking advantage of mouse models with T cell specific functional depletion of these receptors. We aim to translate this finding to the treatment situation of MS relapses. For this planned prospective trial, we collaborate with the Neuroimmunology of the University Hospital Zurich and have submitted a funding application to the Swiss National Foundation (IICT; decision expected in Spring 2022).
Sex differences in neuroimmunological diseases

- Sex differences in MS relapse
In line with the preclinical project on the understanding of steroid signaling, we also clinically investigate MS relapses. A current project focuses on sex-specific differences of relapse phenotypes. Sex differences are mostly only evaluated as a covariate, but sex and gender are not separately analyzed for differential outcomes, a gap of knowledge exists in many aspects of neuroimmunological disorders including MS. The understanding of sex differences, however, is the basis for precision medicine. Up to date, there are no means to predict relapse outcomes. The aim of this project is a multimodal characterization of MS relapses with regards to sex and the identification of potential biomarkers of MS relapse outcome, in the long run to try to find optimized and personalized relapse treatment approaches.
- Sex differences for specific immunotherapies of MS
Combining clinical and lab research, such as a slightly decreased efficacy of sphingosine-1 phosphate (S1P-) inhibitors in young female MS patients, we are modelling different hormonal and S1P situations in the mouse model to study sex differences in more depth aiming to understand how we could improve respective treatments, but also to tackle unmet needs in the field of sex differences in human medicine.
Neuroimmunological Registry (NI-reg)
For our clinical research, we are running a prospective registry including all consenting patients treated at our outpatient neuroimmunological section. With a participation rate of 80-90%, our registry includes more than 1600 neuroimmunological patients. Regarding MS, which is the most common diagnosis, we have a coverage rate of 70-80% for the catchment area of the canton Bern. All patients are routinely included into the liquid biobank Bern (LBB). Main focus of our registry follows the aim to improve treatment in terms of risk-benefit evaluation. Thus, we have gathered clinical data on different effects of S1P treatment between men and women in our NI-reg which is now studied in our basic science laboratory to identify underlying signaling pathways. Merging of our data with open data sources, effects of immunotherapies during the SARS-CoV2 pandemic have been investigated. As one of the main non-motor symptom of MS, but also as a commonly reported symptom after COVID-19, fatigue is currently being investigated to understand and treat this disabling symptom.